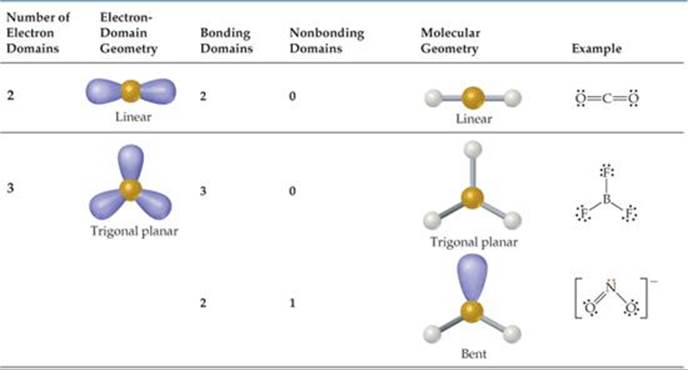 
In contrast, molecular geometry focuses solely on the arrangement of atoms in the molecule. The resulting shape is bent with an H-O-H angle of 104.5°. Key Takeaways Electron geometry describes the spatial arrangement of electron pairs (including bonding and lone pairs) around a central atom in a molecule. Electron group geometries refer to the five geometries: linear, trigonal planar, tetrahedral, trigonal bipyramidal, or octahedral. There are two bonding pairs and two lone pairs. This means there are four electron pairs arranged in a tetrahedral shape. If the central atom does not have any lone pairs (or a single electron), the molecular geometry is the same as the electron pair geometry. The molecular geometry only takes into consideration the number of bonds. In other words, a double bond is one domain, not two.What is the molecular geometry of h2o?Water or H2O has 8 electrons around the central oxygen atom. The electron pair geometry is determined by the total number of charge clouds around an atom. Keep in mind, multiple bonds (i.e., double bonds, triple bonds) count as one electron domain. Use the angular arrangement of the chemical bonds between the atoms to determine the molecular geometry. Similarly one may ask, how do you find the electron domain and molecular geometry? Count the total number of electron domains. Linear - Trigonal Planar - Tetrahedral - Trigonal Bipyramidal - Octahedral Molecular Geometry This is the shape the actual connections between atoms take in a compound. These electrons can be in either a chemical bond or in lone pairs. In the SO2 structure the oxygen are not in lined with each other which means there a positive and negative end. See Below Explanation: Electron Geometry is the shape the electrons take around the central atom. If there are no lone pairs, the electron pair geometry and molecular geometry will. The number of bonding electrons pairs is calculated to determine the molecule’s shape. The total number of electron pairs is calculated to determine the molecule’s shape. It is the 3D arrangement of all the atoms in a particular molecule.Furthermore, why is so2 bent and not linear? CO2 is linear and the SO2 structure is bent, because CO2 has a negative oxygen on each side of the positive carbon they cancel each other out. Molecular Geometry predicts the shape of a molecule by considering bond pairs only. The electron-pair geometries will be the same as the molecular structures when there are no lone electron pairs around the central atom, but they will be different when there are lone pairs present on the central atom. Molecular geometry, on the other hand, helps us understand the entire atom and its arrangement. Hereof, what is the difference between electron domain geometry and molecular geometry?Electron geometry teaches us about the arrangement of different electron groups. Electron domain geometries are based on the total number of electron pairs, while molecular geometries describe the arrangement of atoms and bonding pairs in a molecule.Click to see full answer. 
According to VSEPR, electron pairs distribute themselves around a central atom in such a way as to maximize their distance from each other.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
